 
This is particularly important when the recommended agent is a new and/or infrequently employed drug. However, in view of ongoing research, changes in government regulations, and the constant flow of information relating to drug therapy and drug reactions, the reader is urged to check the package insert for each drug for any changes in indications and dosage and for added warnings and precautions. Drug Dosage: The authors and the publisher have exerted every effort to ensure that drug selection and dosage set forth in this text are in accord with current recommendations and practice at the time of publication. Usage and distribution for commercial purposes requires written permission. Open Access License / Drug Dosage / DisclaimerThis article is licensed under the Creative Commons Attribution-NonCommercial 4.0 International License (CC BY-NC). 
Given the rapid and sustainable efficacy of idarucizumab, the reversal of dabigatran followed by thrombolysis seems to be safe, but further studies and register data are still needed to confirm our preliminary observation, especially to provide additional data concerning the risk-benefit evaluation. To date, the literature lacks data concerning the thrombolytic treatment of acute ischemic stroke in patients after specific reversal of the non-vitamin K oral anticoagulant dabigatran using idarucizumab. Despite the IVT, the patient’s clinical condition deteriorated and she developed an ischemic lesion in the right pons, the right thalamus and right cerebellum. An intracerebral hemorrhage was excluded after systemic thrombolysis. After informed consent, we reversed the anticoagulant effects of dabigatran using idarucizumab and applied an intravenous thrombolysis (IVT) with recombinant tissue plasminogen activator (off-label use).  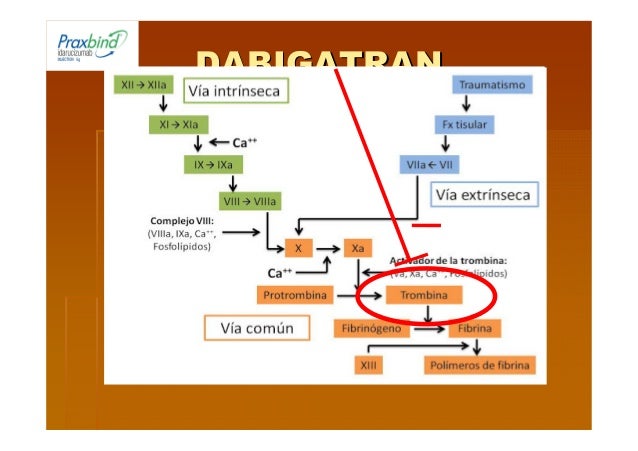
We describe a 75-year-old female patient with nonvalvular atrial fibrillation who presented with acute ischemic stroke during treatment with dabigatran 2 × 110 mg per day. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
